In this post I will talk about rare earth, fundamental elements to modern technologies.
What are?
Rare earth are 17 chemical elements with similar features, they are: yttrium (Y), scandium (Sc) and the lanthanide elements (lanthanum (La), cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu), gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb) and lutetium (Lu)). These elements are highlighted in the periodic table.

Here shows what the elements look like.

This is promethium, radioactive rare earth.

Despite the name, these elements are abundant in nature, the term comes from a time where it believed they were rare. In nature, the most common ores are:
- Monazite whose formula is (Ce,La,Nd,Th,Y,Sm)PO_{4}, it has cerium, lanthanum, yttrium, neodymium and samarium. The most common type is enriched with cerium. The figures below show types of monazite ore which possess rare earth.

CePO_{4}

LaPO_{4}

NdPO_{4}

SmPO_{4}
- Bastnasite whose formula is (Ce,La,Y)CO_{3}F, it has cerium, lanthanum and yttrium. Below are the types of this ore.

CeCO_{3}F

LaCO_{3}F

YCO_{3}F
- Xenotime, the most common type is composed by yttrium, but have dysprosium, erbium, terbium and ytterbium ores.

YPO_{4}

YbPO_{4}
- Loparite whose formula is (Na,Ce,Sr)(Ce,Th)(Ti,Nb)_{2}O_{6}, the most part has cerium.

Extraction and process
It is very expensive and complicated to extract rare earth. First the mineral is crushed until gravel size, then it is milled in a mill. One of the methods to extract rare earth from minerals consist in dissolve the ore in an acid solution and put it in the mixer to separate by weight depending on the acidity.

Other separation method consist in use ion exchange chromatography, I will explain with more details in a post about this type of chromatography.

After the separation process, rare earth stays in oxide form, to be transformed in pure metal.

The figure below shows the entire process’s flowchart.
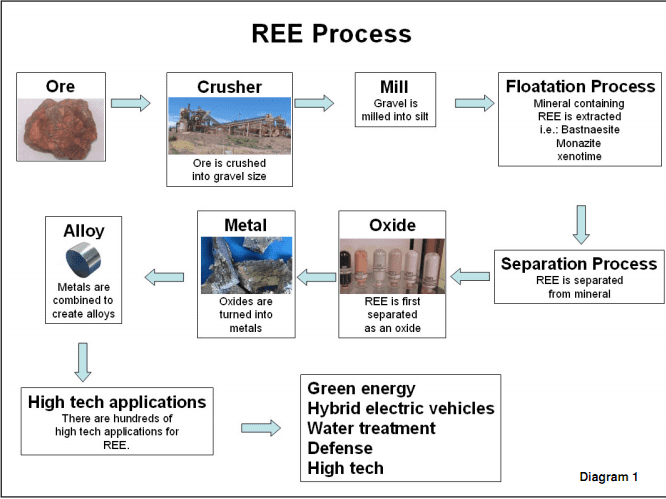
In addition to the environmental impact caused by the mining activity. During this process showed above, undesirable minerals are discarded and the processing plants generate many toxic and radioactive waste which poison water and farms.

In the next post about environment, I will talk where are the greatest rare earth reserves and about each element and your applications.


Superb posts! Have a look at my page webemail24 where I also put in extra effort to create quality information about Health Supplements.
That link is about e-mail marketing.
Thank you all.
Some truly prime posts on this internet site , saved to bookmarks .
I regard something truly special in this internet site.
I’m extremely pleased to discover this great site. I want to to thank you for ones time just for this fantastic read!! I definitely loved every little bit of it and I have you saved as a favorite to look at new information in your web site.
Over and over again I like to think about this problems. As a matter of fact it wasn’t even a month ago that I thought about this very thing. To be honest, what is the answer though?
I just want to say I’m new to blogging and site-building and really loved your web site. Very likely I’m planning to bookmark your website . You certainly have amazing article content. Thanks for sharing your web site.